Fast Calculation of van der Waals Volume as a Sum of Atomic and Bond Contributions and Its Application to Drug Compounds | The Journal of Organic Chemistry

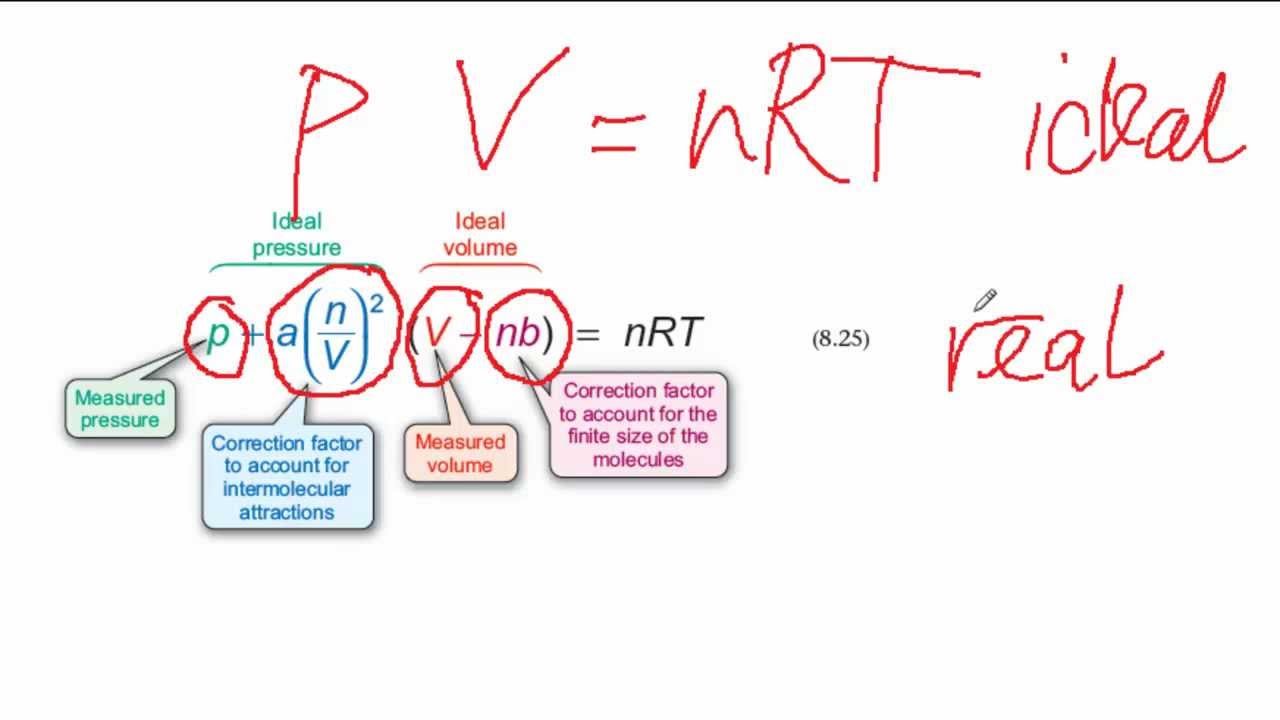
Using van der Waals equation, calculate the constant `a` when `2 mol` of a gas confined in - YouTube

Using van der Waals equation, calculate the constant a when 2 mol of a gas confined in a 4 L flasks exerts a pressure of 11.0 atm at a temperature of 300

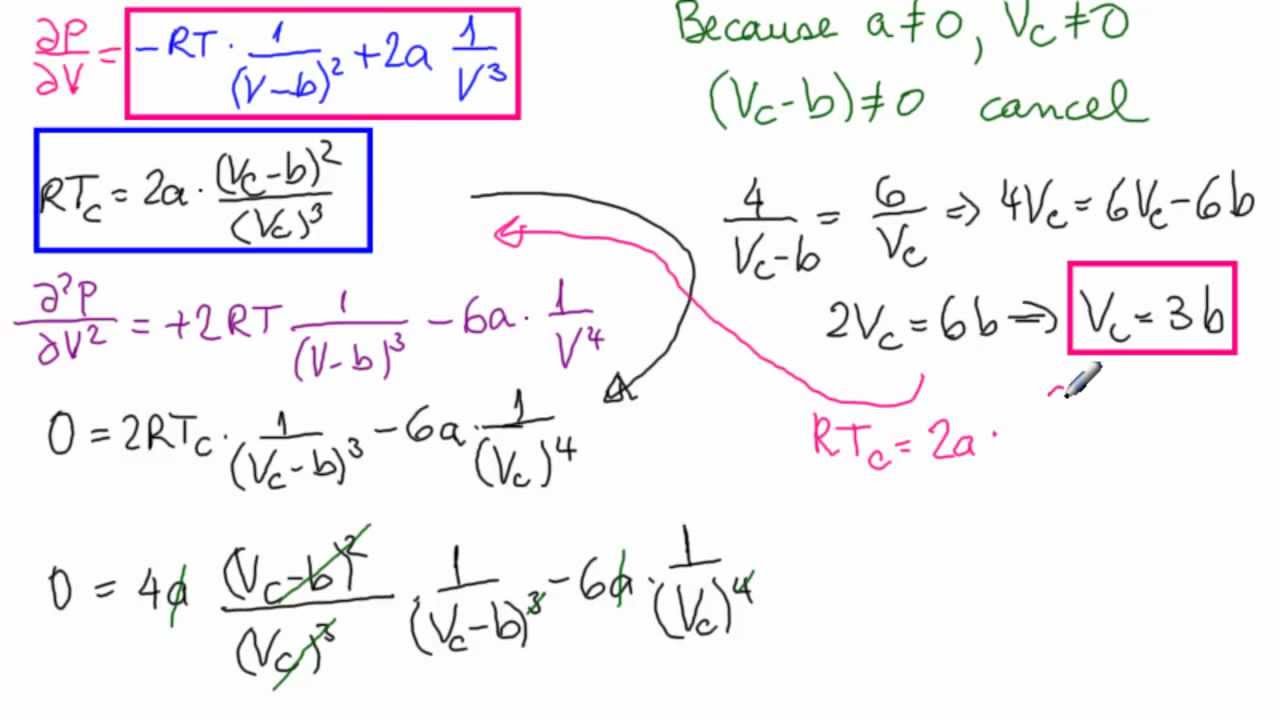
SOLVED: 514. Calculate the pressure exerted by L.OO mol of COz in a L.00 L vessel at 300 K assuming that the gas behaves ideally: (6) Repeat the calculation using the van

SOLVED:Use both the ideal gas equation and the van der Waals equation to calculate the pressure exerted by 1.50 mol of SO2(g) when it is confined at 298 K to a volume
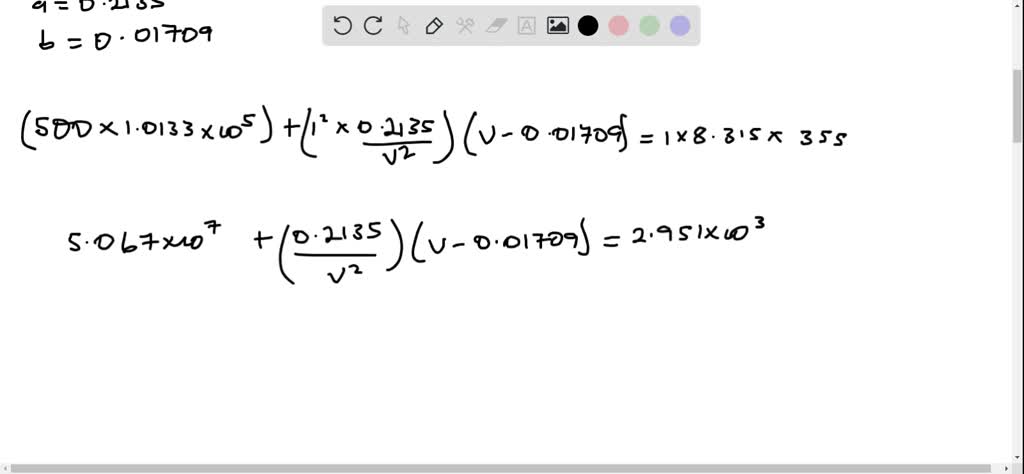
SOLVED: Use the van der Waals equation and the ideal gas equation to calculate the volume of 1.000 mol of neon at a pressure of 500.0 atm and a temperature of 355.0