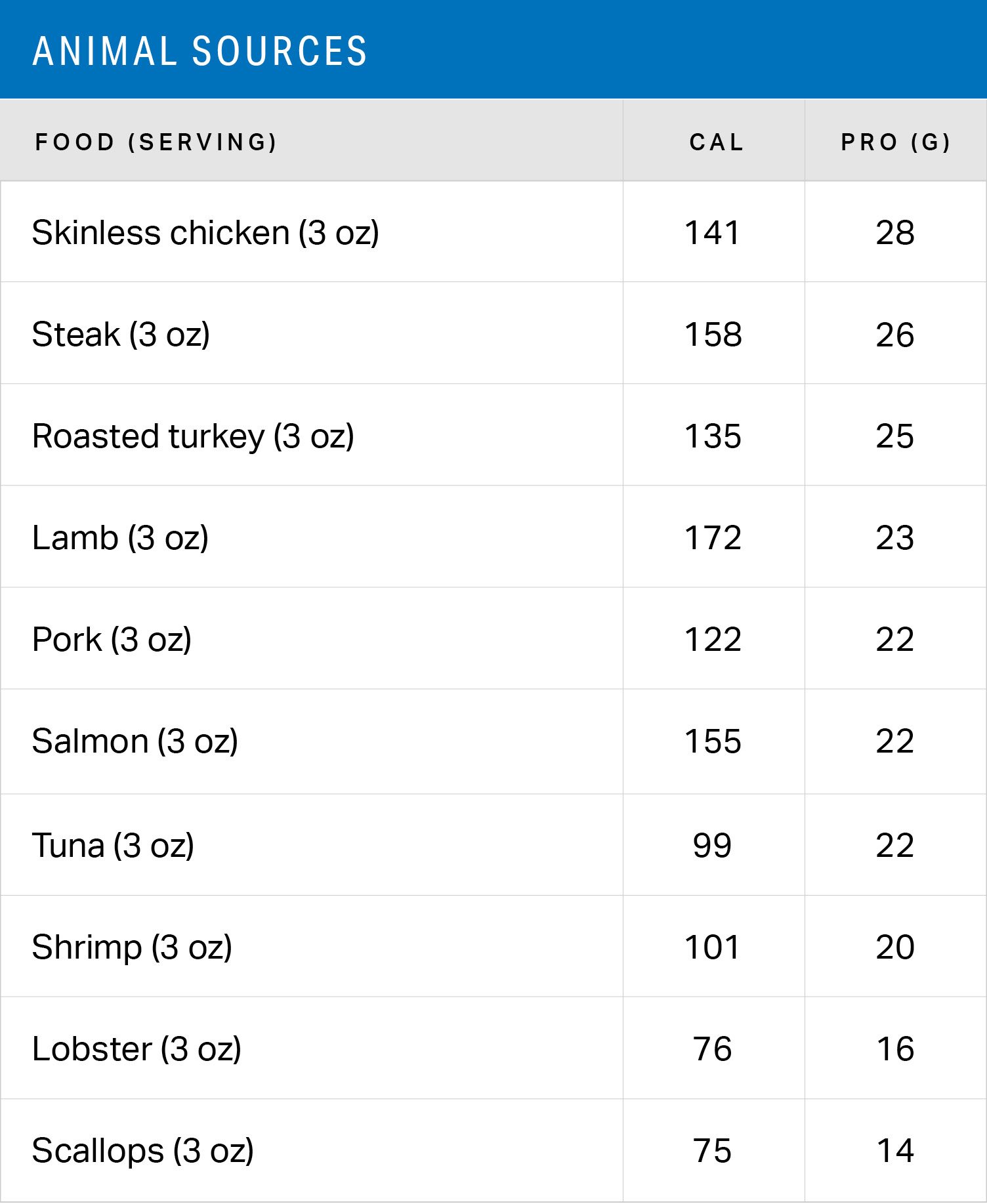
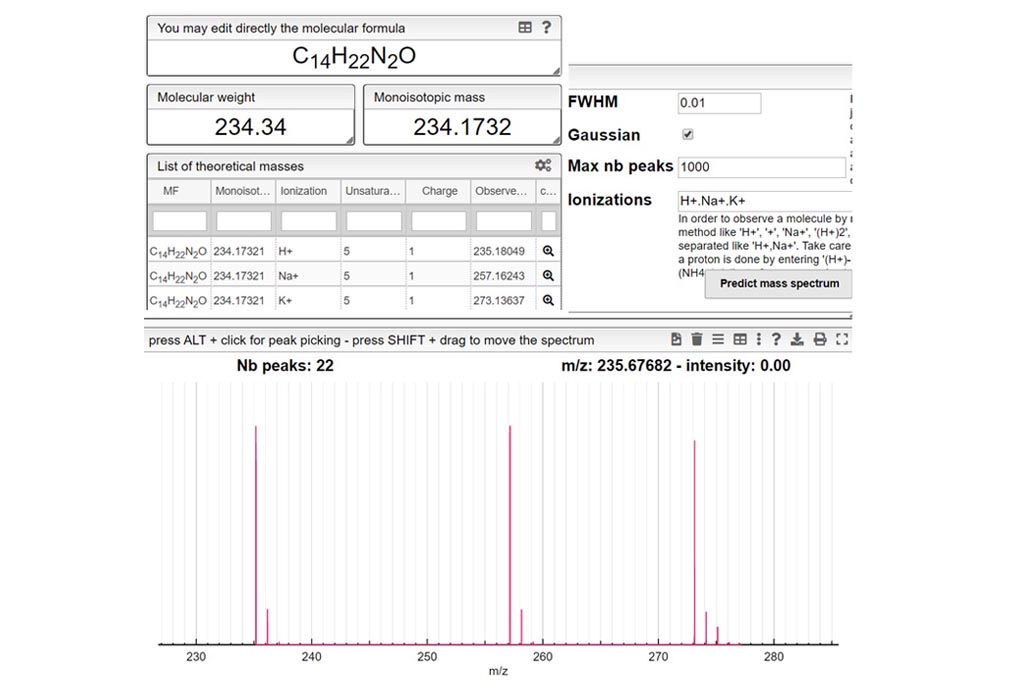
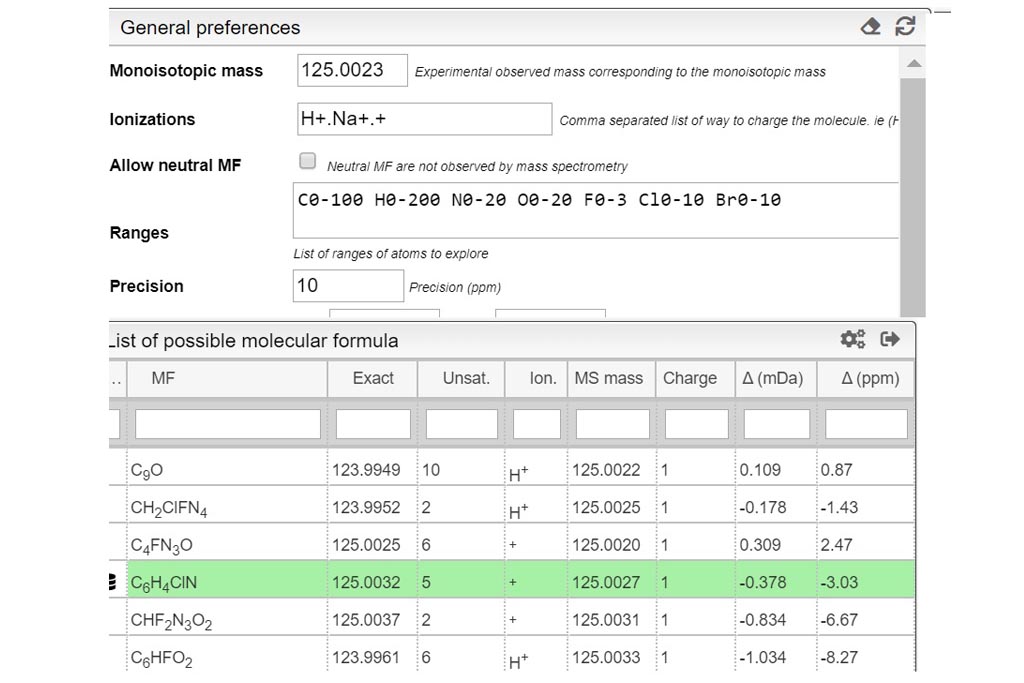
POTAMOS mass spectrometry calculator: Computer aided mass spectrometry to the post-translational modifications of proteins. A focus on histones - ScienceDirect

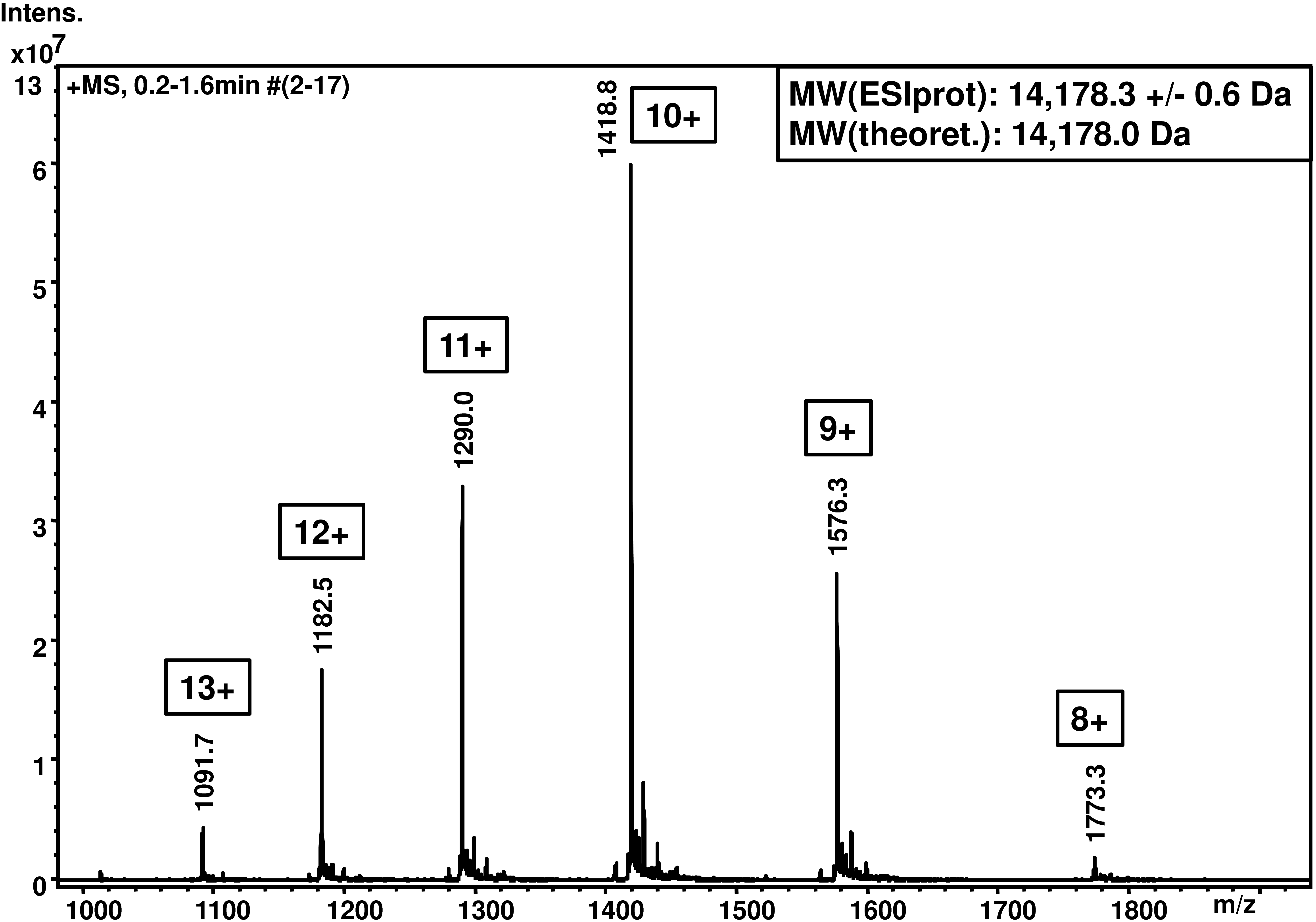
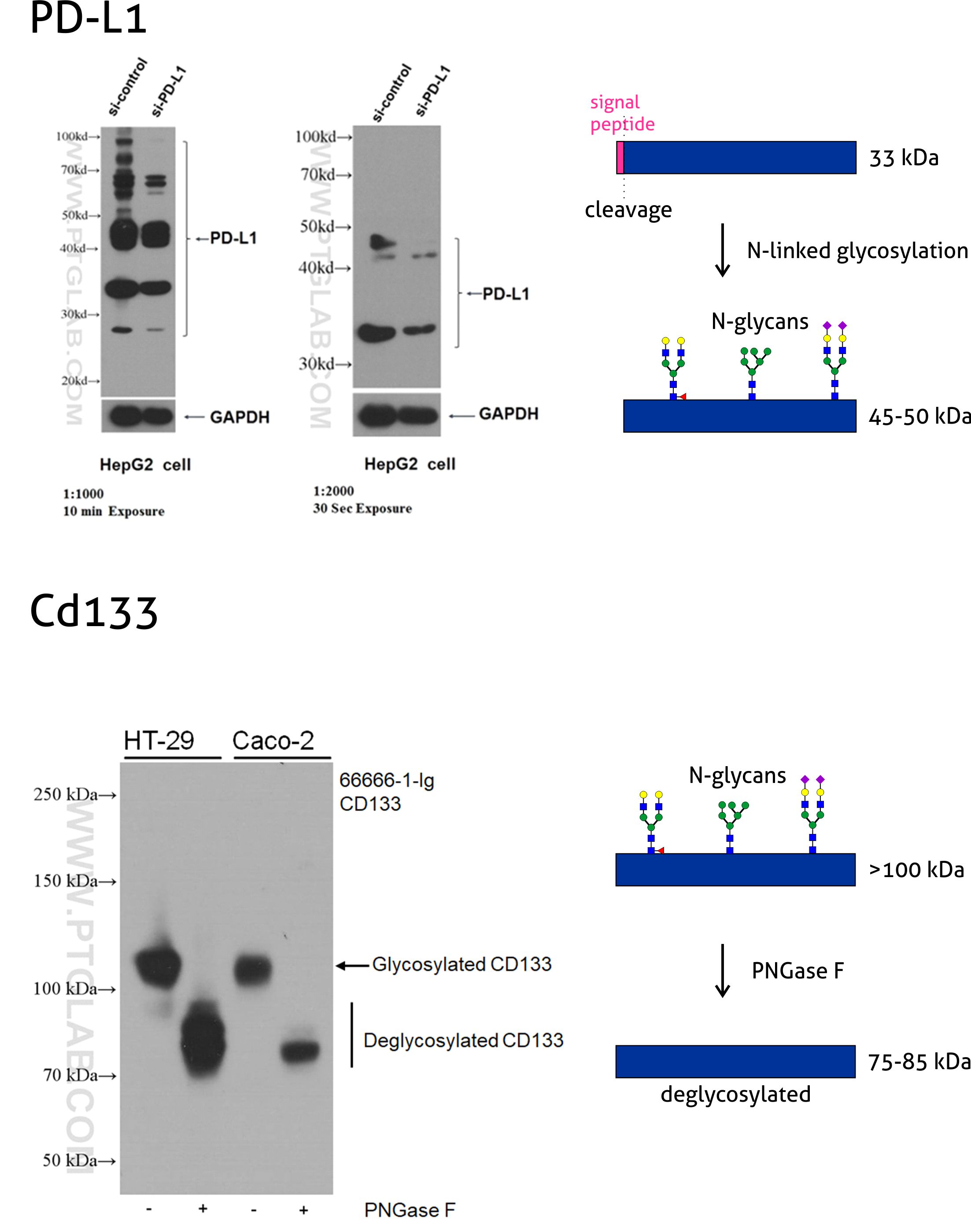
Why Does the Molecular Weight of My Protein Differ From the Theoretically Expected Weight? | Technology Networks

SAXSMoW 2.0: Online calculator of the molecular weight of proteins in dilute solution from experimental SAXS data measured on a relative scale - Piiadov - 2019 - Protein Science - Wiley Online Library

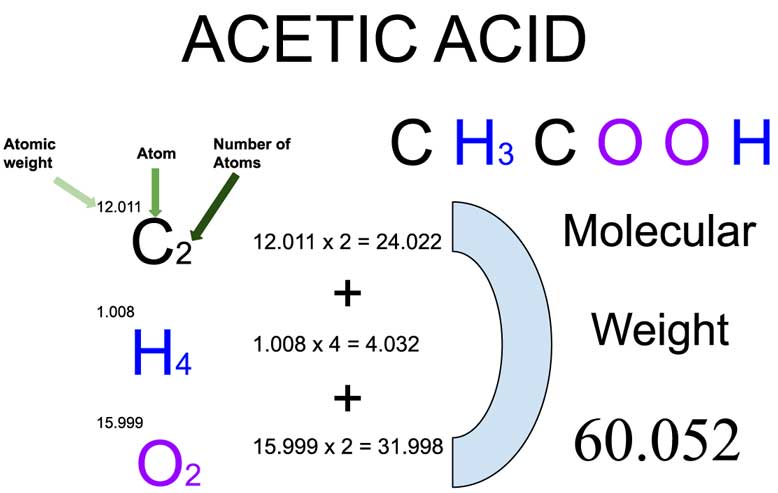





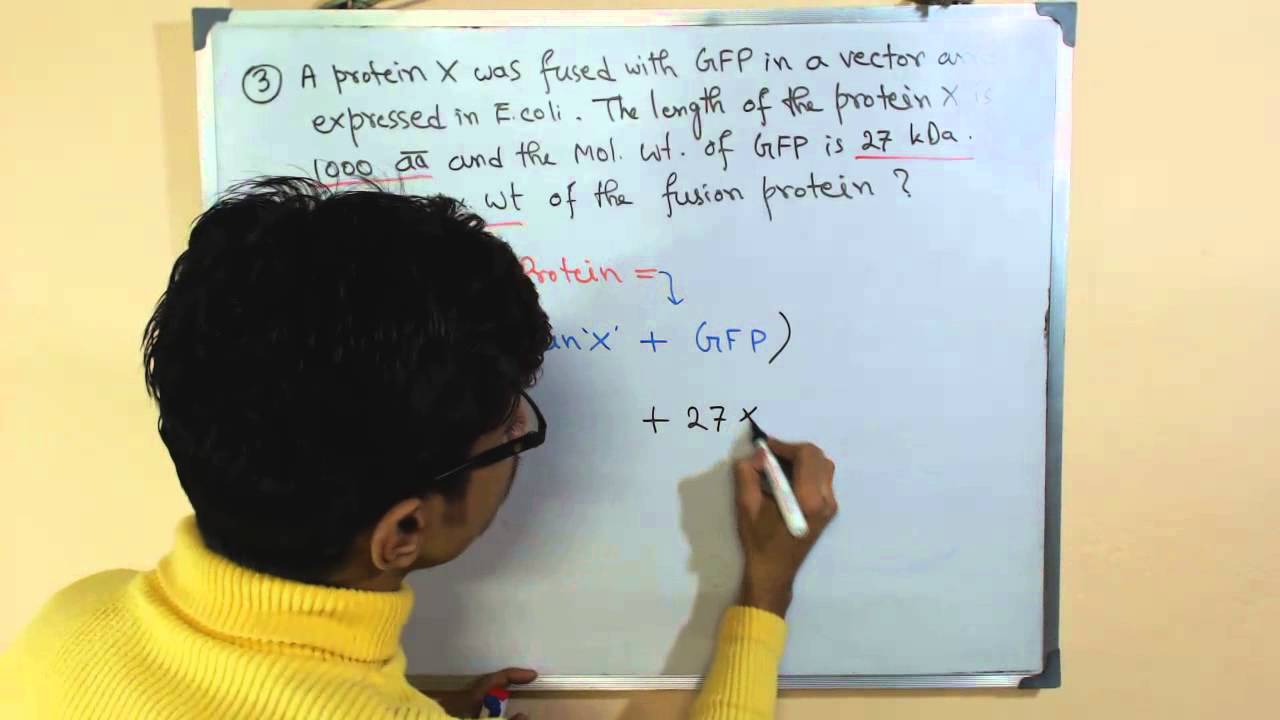


![Glycine [C2H5NO2] Molecular Weight Calculation - Laboratory Notes Glycine [C2H5NO2] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2022/06/glycine-molecular-weight-calculation.jpg)